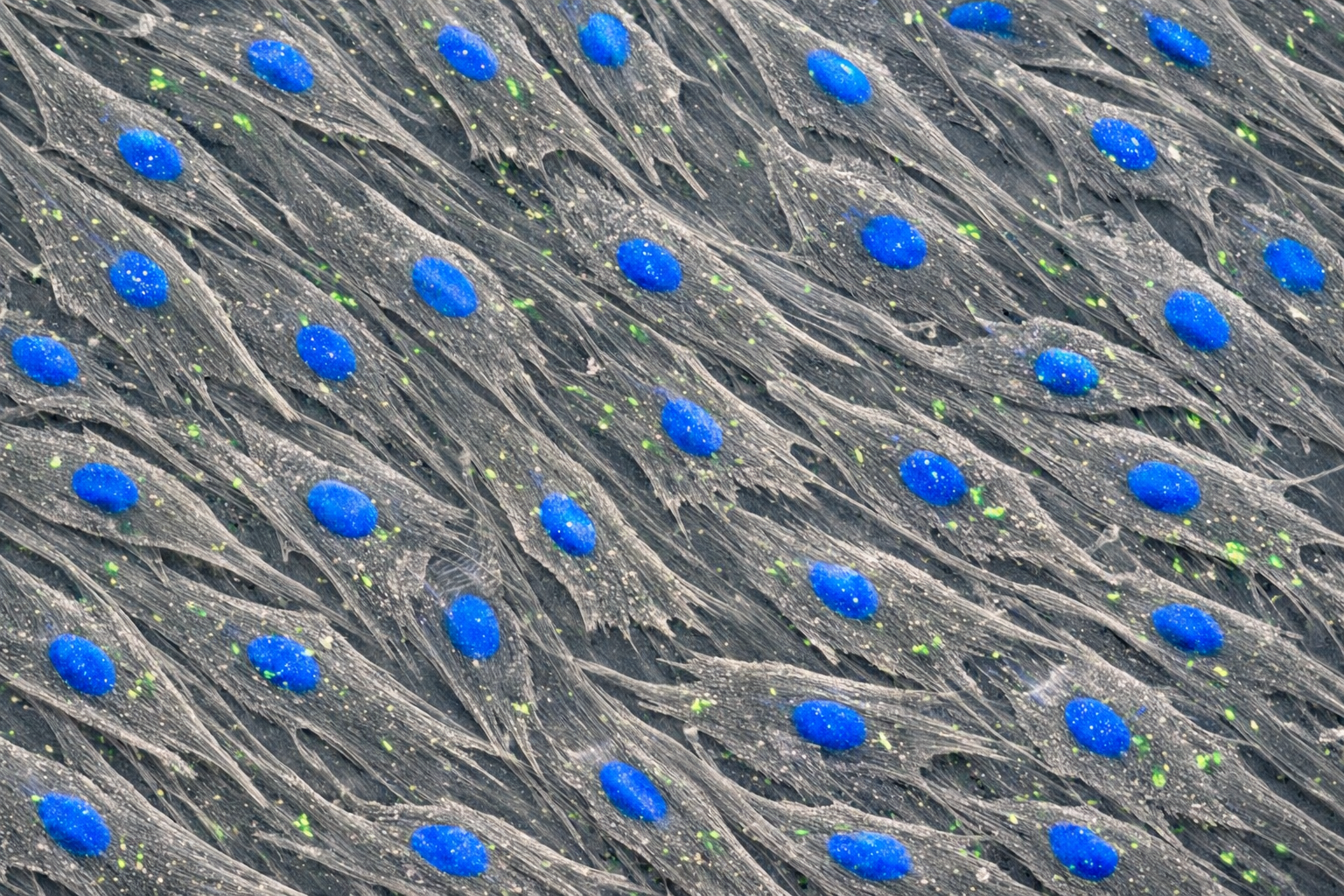
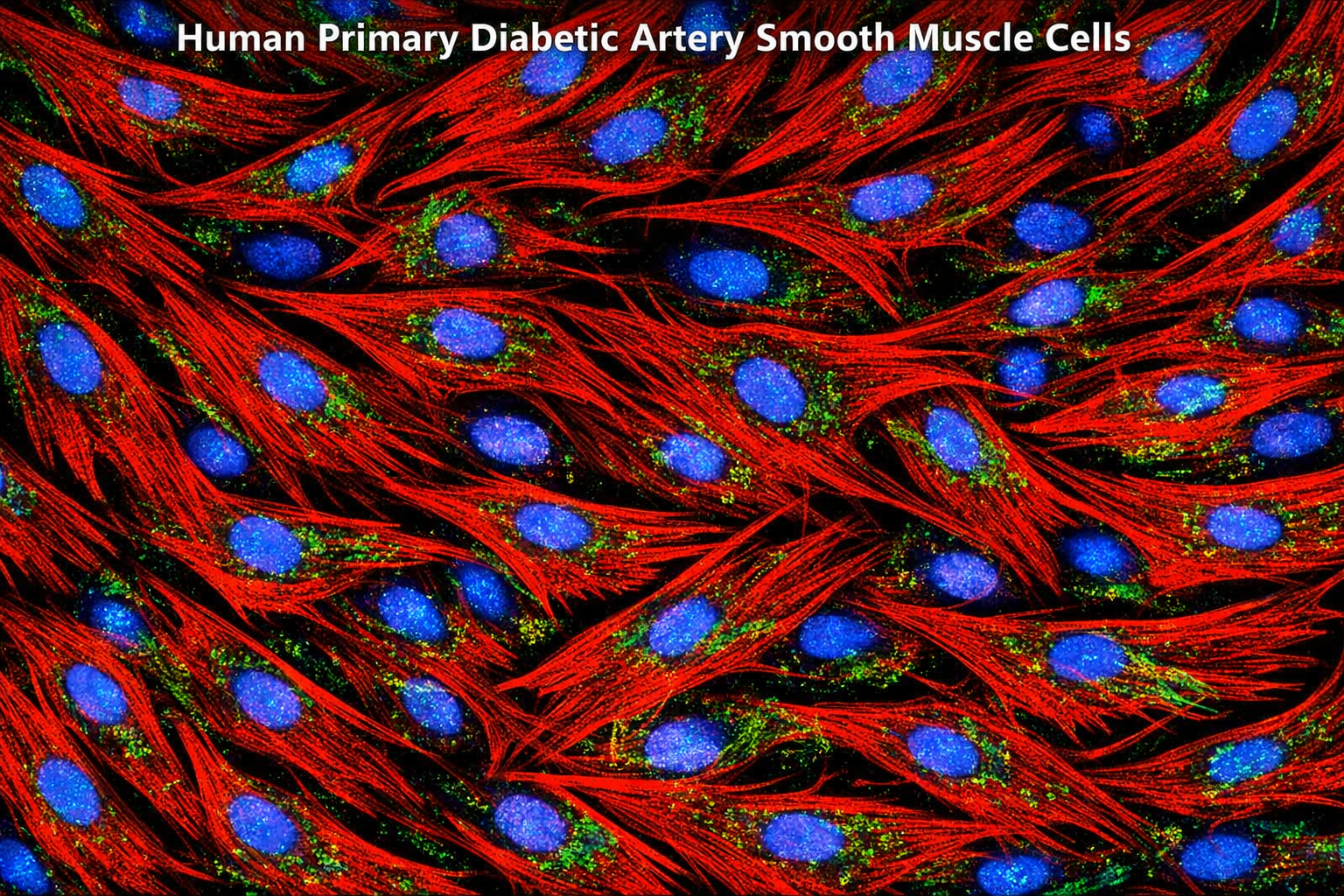
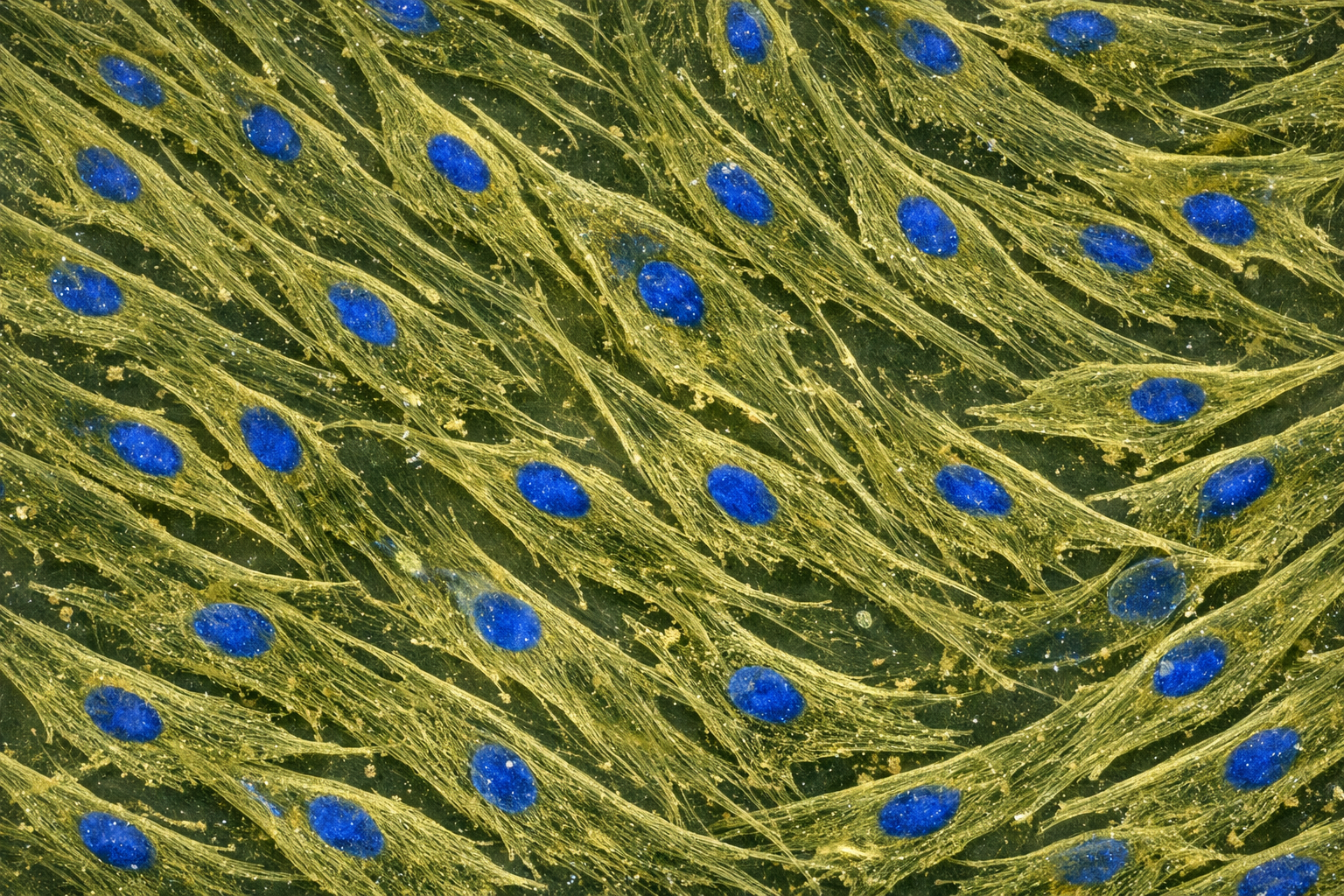
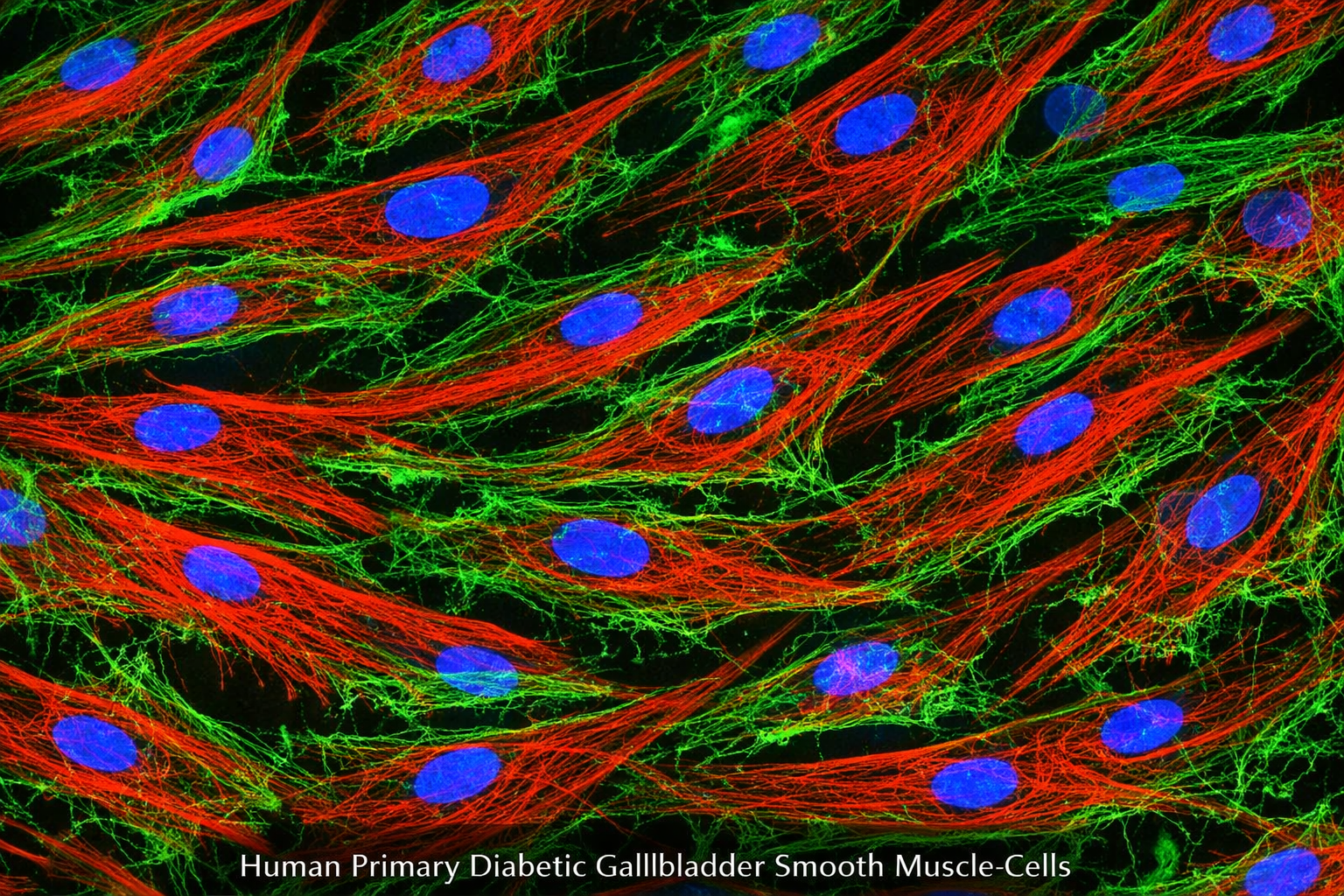
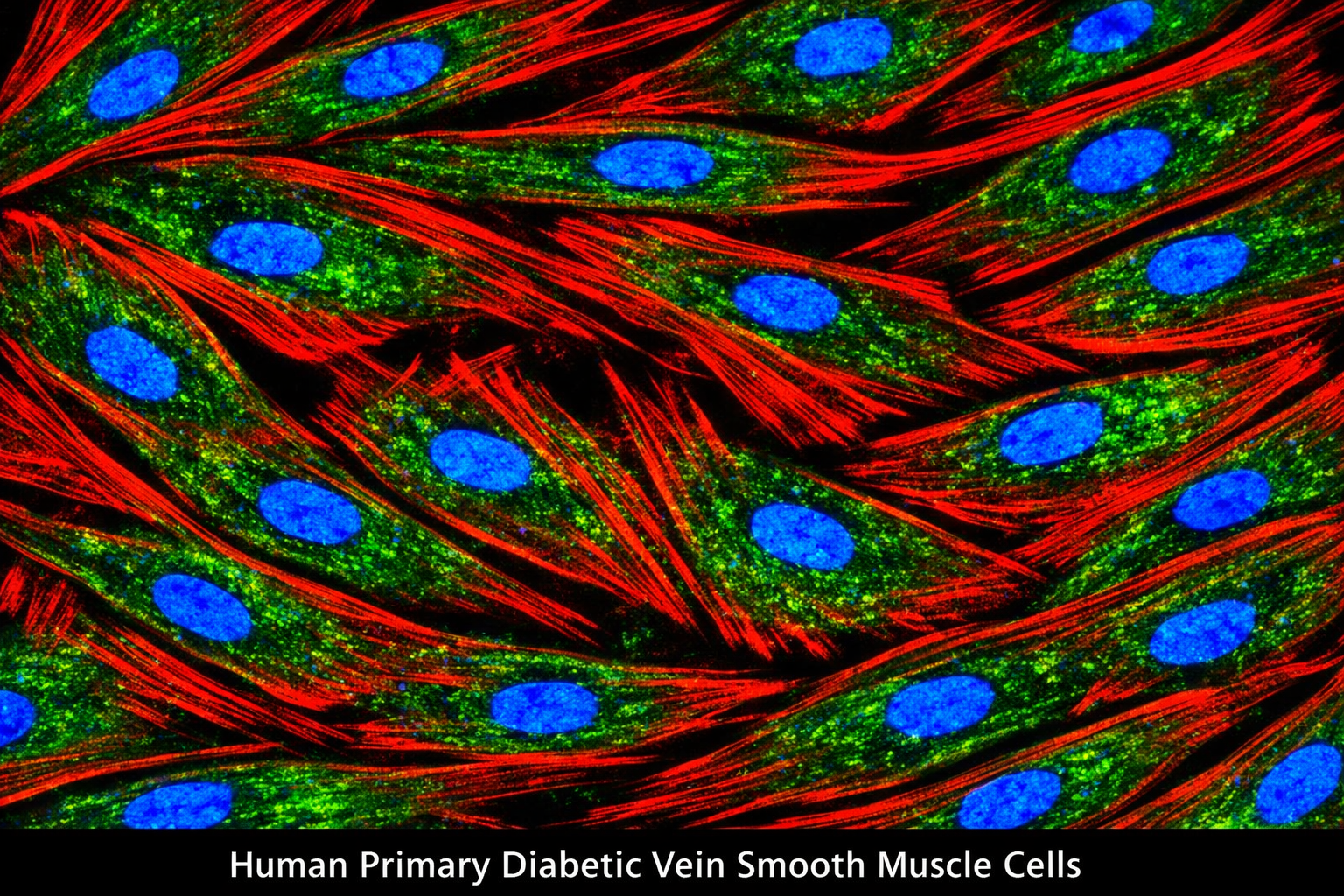
Human Primary Diabetic Vein Smooth Muscle Cells
BVXDS013
Certified
QualityGlobal
ShippingSupport
24/7Info
IncludedProduct Details
Size
1ml
Product Code
BVXDS013
Quick Specifications
- Product Type: Human Type II Diabetes Endothelial Cells
- Viability: >95% viable (Trypan Blue exclusion)
- Storage: Store in liquid nitrogen vapor phase (-196°C) for long-term. Ship on dry ice (-80°C).
- Shelf Life: Very stable in the Liquid Nitrogen Vapor Phase
- Catalog: BVXDS013
- Growth: Primary cells capable of multiple passages when using recommended medium and proper handling
Human Primary Diabetic Vein Smooth Muscle Cells
Product Description
Human Primary Diabetic Vein Smooth Muscle Cells are a specialized type of cell line derived from human veins affected by diabetes. These cells exhibit unique characteristics that reflect the physiological and pathological conditions associated with diabetes, particularly in relation to vascular health. They are essential for studying the effects of diabetes on vascular smooth muscle function, signaling pathways, and potential therapeutic interventions aimed at improving cardiovascular outcomes in diabetic patients. Guarantee expansion for a minimum of 2-5 population doublings. IMPORTANT: Quality warranty requires use of Bioverax media. Biohazardous material despite negative pathogen testing. Quality claims must be reported within 1 month.
Key Features & Benefits
Catalog Number: BVXDS013 Product Format: Frozen Vial Cell Number: > 5x10[5] cells/vial Passage:1(P1) Storage: Liquid Nitrogen, Vapor Phase Intended Use: Research use only (RUO) Characterization Markers Characterized by positive expression of smooth muscle cell-specific alpha-actin.
Quick Facts
Product Type
Human Type II Diabetes Endothelial Cells
Viability
>95% viable (Trypan Blue exclusion)
Storage
Store in liquid nitrogen vapor phase (-196°C) for long-term. Ship on dry ice (-80°C).
Shelf Life
Very stable in the Liquid Nitrogen Vapor Phase
Applications
Technical Specifications
| Product Type | Human Type II Diabetes Endothelial Cells |
|---|---|
| Viability | >95% viable (Trypan Blue exclusion) |
| Proliferative Capability | Primary cells capable of multiple passages when using recommended medium and proper handling |
| Storage Conditions | Store in liquid nitrogen vapor phase (-196°C) for long-term. Ship on dry ice (-80°C). |
| Shelf Life | Very stable in the Liquid Nitrogen Vapor Phase |
| Product Code | BVXDS013 |
| Size/Format | 1ml |
| Category | Primary Cells |
Primary Applications
Applications & Use Cases
Primary Applications
Restenosis and Vein Graft Failure: Diabetic patients have significantly higher rates of vein graft occlusion after bypass surgery. Researchers use these cells to test drugs (e.g.
Suitable for restenosis and vein graft failure: diabetic patients have significantly higher rates of vein graft occlusion after bypass surgery. researchers use these cells to test drugs (e.g. studies and research applications
mTOR inhibitors
Suitable for mtor inhibitors studies and research applications
anti-proliferatives) that might prevent the excessive smooth muscle cell proliferation that closes off the graft. Diabetic Vasculopathy: Understanding why diabetic patients suffer from accelerated atherosclerosis and peripheral artery disease (PAD) involving the venous system. Wound Healing: Venous insufficiency is a major cause of diabetic foot ulcers. Studying the contractility and migration of these cells helps understand why wound contraction fails in diabetic patients. Drug Screening: Pharmaceutical companies use these primary cells to test the vascular toxicity or efficacy of new antidiabetic drugs
Suitable for anti-proliferatives) that might prevent the excessive smooth muscle cell proliferation that closes off the graft. diabetic vasculopathy: understanding why diabetic patients suffer from accelerated atherosclerosis and peripheral artery disease (pad) involving the venous system. wound healing: venous insufficiency is a major cause of diabetic foot ulcers. studying the contractility and migration of these cells helps understand why wound contraction fails in diabetic patients. drug screening: pharmaceutical companies use these primary cells to test the vascular toxicity or efficacy of new antidiabetic drugs studies and research applications
ensuring that therapies intended to lower glucose do not inadvertently cause vascular smooth muscle proliferation.
Suitable for ensuring that therapies intended to lower glucose do not inadvertently cause vascular smooth muscle proliferation. studies and research applications
Research Use Cases
In Vitro Studies
Cell culture and screening applications
Molecular Biology
Gene expression & signaling studies
Drug Discovery
Screening & validation studies
Technical Support & Documentation
Technical Support
Our experts are available for technical questions and protocol assistance.
Contact Support